Background
Acute lymphoblastic leukemia (ALL) is the most common leukemia found in children. Chimeric Antigen Receptor (CAR) T cells induce high response rates against relapsed/refractory B-cell ALL, but 40% of CAR-T recipients suffer disease relapse, many due to poor in vivo survival of the CAR-T cells. Metabolic exhaustion has been proposed as a major barrier to optimal CAR-T cell performance. Therefore, modulating T cell metabolism may represent a promising method to improve CAR T-cell therapy. AMP-activated protein kinase (AMPK) is a heterotrimeric signaling complex which serves as a cellular energy sensor, promoting mitochondrial health and oxidative metabolism under conditions of energetic stress. We have demonstrated overexpressing the regulatory subunit AMPKγ2 increases AMPK signaling in human T cells, and hypothesized that this overexpression would enhance CAR-T metabolic fitness and improve anti-leukemia activity.
Results
We used lentiviral transduction to introduce a CD19-reactive, CD28 CAR into human T cells, followed by transduction of either AMPKγ2 or an empty vector (EV) control. Dual transduced cells were isolated by flow sorting and cultured with human IL2.
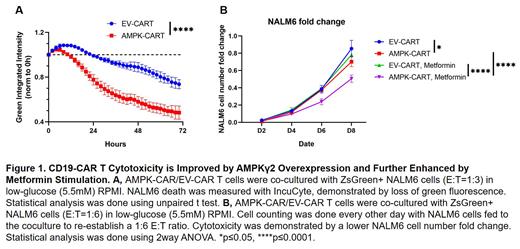
The metabolism and cytotoxicity of these CAR-T cells were first assessed in vitro. AMPK-CART cells showed higher oxidative metabolism, with a 29% increase in basal oxygen consumption rates (OCR) and a 45% increase in maximal OCR (p<0.001) following overnight stimulation by NALM6 cells. In addition, co-culture with Zs-Green+ NALM6 cells revealed greater cytotoxicity by AMPK-CART cells (p<0.001) (Fig. 1A), accompanied by a 35% increase in CD25 expression (p<0.05). We then pivoted to studying AMPK-CART cells in vivo utilizing a murine xenograft leukemia model. Interestingly, despite their in vitro advantages, AMPK-CART cells demonstrated equivalent anti-tumor capacity in vivo.
We reasoned that AMPKγ2 overexpression may simply protect AMPK signaling which is already occurring. To test this idea, we coupled AMPKγ2 overexpression with transient metformin treatment, a process known to activate AMPK signaling through inhibition of mitochondrial complex I. Metformin treatment was followed by the functional analysis of CAR-T cells in a chronic stimulation protocol where we repetitively stimulate CAR-T cells with NALM6 targets to induce exhaustion. After 8 days of chronic stimulation, metformin treated AMPK-CART cells (7mM for 4 hours) demonstrated the highest cytotoxicity against NALM6 cells compared to either metformin treated EV-CART cells or AMPK-CART cells without metformin treatment (Fig. 1B). This cytotoxic advantage was accompanied by an 18% increase in CD25 expression (2052.67±104.74 vs. 1742.67±14.64, p<0.05) and a 56% elevation in the percentage of central memory cells (9.15±1.32 vs. 5.88±0.25, p<0.05) in metformin treated AMPK-CART cells. These changes were not seen in EV-CART cells with metformin treatment.
Conclusions
Here, we report that AMPKγ2 overexpression in CD19-CAR T cells enhances oxidative metabolism and improves in vitro cytotoxicity but does not significantly increase in vivo anti-leukemia function. Metformin pre-treatment elevates CAR-T cytotoxicity following chronic antigen stimulation, but only when coupled with AMPKγ2 overexpression. This combination is what we predict will improve the function of CD19-CAR T cells in our murine xenograft leukemia model and human samples.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal